Case #008 - The Review That Put a Bestselling Supplement Under Investigation
A customer reported adverse effects, and Amazon gave the seller 30 days to prove manufacturing compliance or lose the listing.
If you’re new here, welcome.
If you’ve been reading for a while, thank you for being here.
Each week, we break down one real Amazon case from the field. Not to share tactics, but to decode how Amazon’s system actually behaves and what to do when it breaks.
All past cases live in a single searchable archive, built to help you identify recurring patterns across time.
Context
The customer feedback that triggers a safety investigation
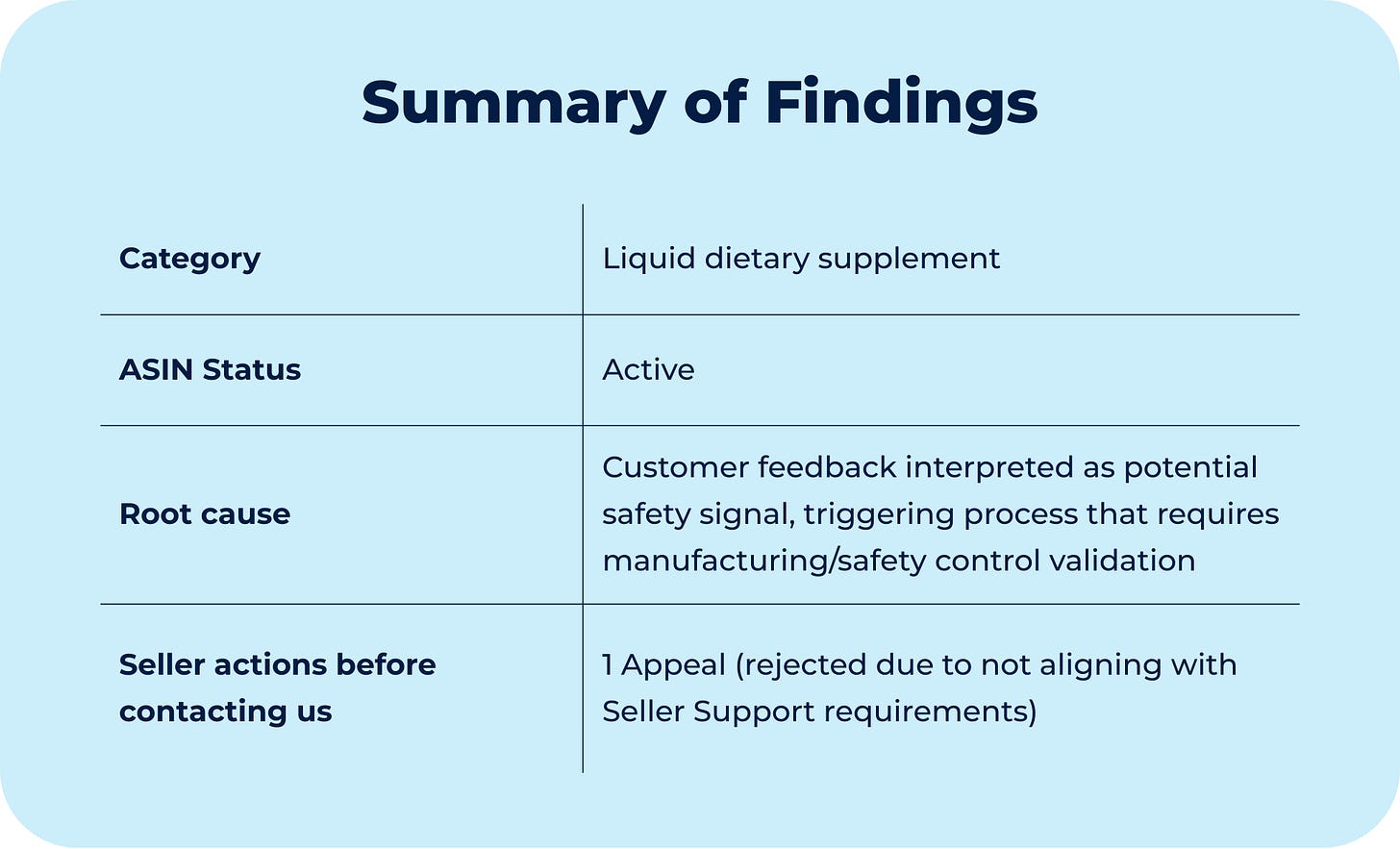
A seller received a Product Safety investigation notice related to a liquid dietary supplement in the herbal wellness category.
Amazon notified the seller that the listing was at risk of deactivation due to potential safety concerns, triggered by customer feedback.
The investigation was initiated after Amazon flagged a customer review describing a possible adverse health effect. In the feedback, the customer stated that while undergoing chemotherapy, they experienced blood clotting issues and attributed recurring bleeding episodes to the use of the product (yes, it was a real deal).
The review explicitly framed the effect as blood thinning, warning other customers of a possible side effect. Based on that feedback, Amazon classified the case as a Food Safety Investigation and requested additional documentation to confirm that the product met current safety standards.
Amazon’s communication made several points clear:
The listing was not yet deactivated, but deactivation would occur if the investigation was not resolved within 30 days.
The investigation was scoped to product safety, not labeling accuracy or restricted claims.
Amazon requested submission of HACCP, GMP, or GFSI certification, or an explanation if such documentation was not available.
The notice emphasized that failure to provide the requested documentation would result in:
Closure of the case
Deactivation of the impacted ASIN
Potential account-level enforcement if unresolved
At this stage, the seller was not being accused of selling an unsafe product. The issue was that Amazon had received customer-reported health feedback significant enough to trigger a formal safety review, shifting the burden to the seller to demonstrate compliance with Amazon’s food and supplement safety requirements.
Diagnostic
Why Amazon escalated a customer review into a safety investigation
Amazon’s Product Safety and Food Safety systems continuously monitor reviews for language that suggests adverse health effects, especially when those effects involve serious medical conditions, drug interactions, or bodily functions such as bleeding, clotting, or organ impact.
In this case, the customer explicitly referenced:
Chemotherapy treatment
Difficulty with blood clotting
Recurrent bleeding episodes
A perceived temporal link between product use and symptoms
That combination of factors placed the feedback into a category Amazon treats as high-risk, regardless of whether the claim can be medically substantiated.
Once flagged, the case was routed under Food Safety Investigation, not under Restricted Products or Dietary Supplements claims enforcement. That distinction matters. Amazon was not evaluating whether the listing content made prohibited claims. It was evaluating whether the product’s manufacturing and safety controls met Amazon’s requirements in light of reported customer harm.
Under Amazon’s Food Safety Investigation policy, when a potential safety issue is raised through customer feedback, the seller is required to demonstrate that:
The product is manufactured under recognized safety standards
Appropriate preventive controls are in place
This is why Amazon requested HACCP, GMP, or GFSI certification, rather than:
A rewritten listing
A clarification of the review
A rebuttal of the customer’s experience
The diagnostic conclusion is that the ASIN was placed under review because customer feedback was interpreted as a potential safety signal, triggering a process that focuses on manufacturing and safety controls.
In cases like this, the hardest part is rarely the fix itself. It’s knowing where the problem actually lives before you start retrying actions that the system is designed to ignore.
When safety investigations are triggered by customer feedback, understanding that Amazon evaluates manufacturing controls (not medical causality) determines whether your response gets the investigation closed or the listing deactivated.
Though Process
How we focused on what Amazon actually needed
In this case, there were three “logical” paths forward:
A) Contextualize the case to Amazon, explaining that the product warned and prevented inappropriate consumption (didn’t work).
B) Provide the requested documents for the investigation, since Amazon is explicitly requesting, and it is mandatory.
C) Use the information to your advantage and give the Amazon system what it actually needs to understand that everything is in order.
To find the best solution we had to focus on the following:
Insight #1: Amazon’s investigation framework validates safety controls, not specific credential formats
Amazon’s Food Safety Investigation process is designed to verify that products meet recognized manufacturing and safety standards when risk indicators appear. The customer review functions as a trigger, it moves the ASIN into a verification workflow, where Amazon evaluates whether the seller operates under controls it recognizes as sufficient.
The question becomes: are GMP/HACCP/GFSI certifications the only artifacts that satisfy this verification requirement, or do they represent examples of what Amazon’s reviewers are trained to evaluate?
These certifications are externally audited, standardized safety programs. They provide pre-validated evidence that a recognized third party has confirmed manufacturing controls. That’s why Amazon trusts them, so internal teams can quickly verify without an independent investigation.
But certifications aren’t always immediately available. GMP certification, for example, requires working with an official certifying body and can take months to obtain. If a seller doesn’t already have this documentation when the investigation opens, obtaining it within the 30-day window may not be operationally feasible.
Insight #2: Amazon used to provide a questionnaire that revealed what they actually evaluate
Approximately a year ago, Amazon’s Food Safety investigation notices included a supplementary questionnaire alongside the certification request. The questionnaire asked structured questions about:
Whether the ASIN had been recalled in the past twelve months
The reason for the reported health issue
Corrective actions were taken to address the specific incident
Preventive actions were implemented to prevent recurrence
This questionnaire is mapped directly to what Amazon’s Food Safety reviewers need to verify: that the seller has identified the risk, understands what controls are needed, and has implemented measures to manage that risk going forward.
Amazon stopped including this questionnaire in its investigation notices. But the underlying review logic didn’t change. The questionnaire revealed what Amazon’s internal process evaluates, and those evaluation criteria still exist even when the questionnaire isn’t explicitly provided.
Insight #3: A Food Safety Plan addresses the same verification framework
A Food Safety Plan is a documented system that identifies potential hazards in the production process and establishes controls to prevent, eliminate, or reduce those hazards to acceptable levels. It’s operationally similar to what GMP certification validates, but it’s a seller-controlled document rather than a third-party credential.
When paired with the questionnaire Amazon used to provide, a Food Safety Plan answers the same questions Amazon’s reviewers need resolved:
What controls are in place during manufacturing?
How does the seller identify and manage food safety risks?
What corrective and preventive actions exist for the specific product under investigation?
This combination of a Food Safety Plan + questionnaire provides structured verification data that maps to Amazon’s internal review checklist, even though it’s not the certification Amazon explicitly requested.
That analysis leads to a simple principle that guided the solution: when Amazon triggers a safety investigation, the winning move is not “argue harder,” it’s match the evidence type Amazon’s process is designed to accept.